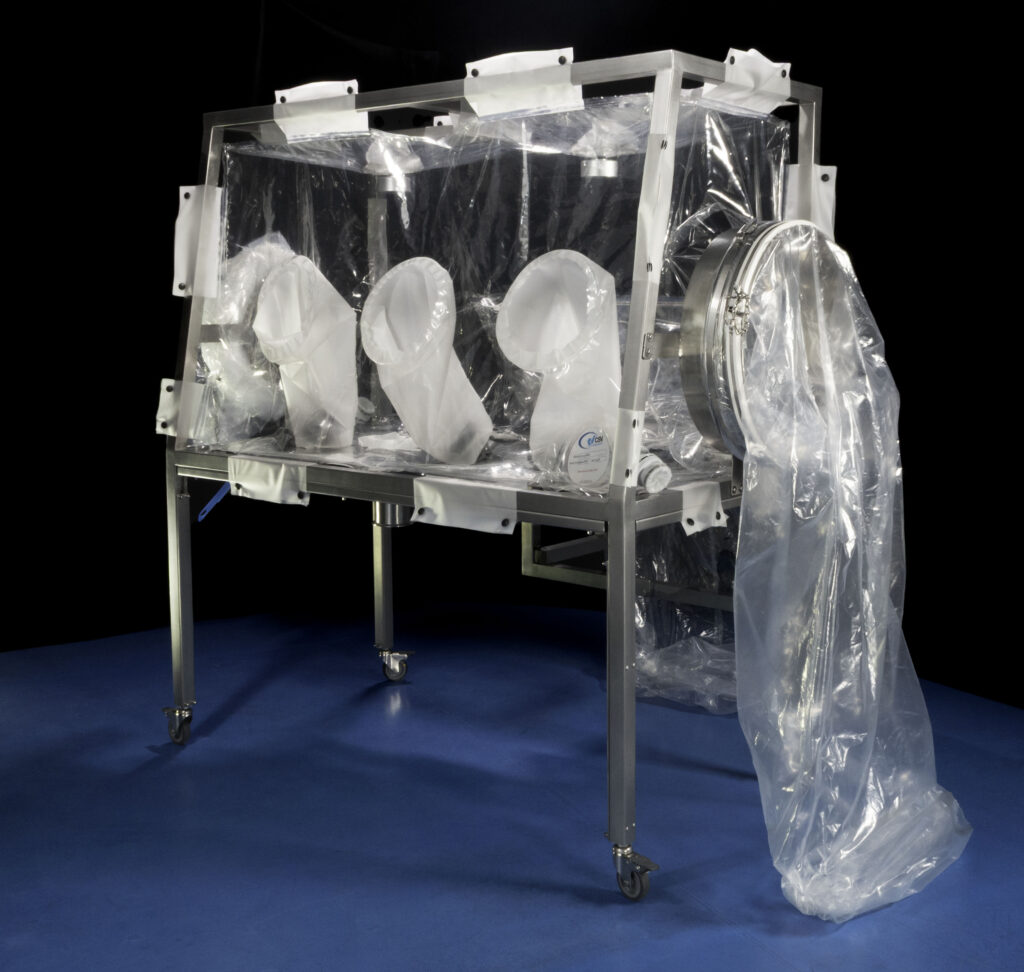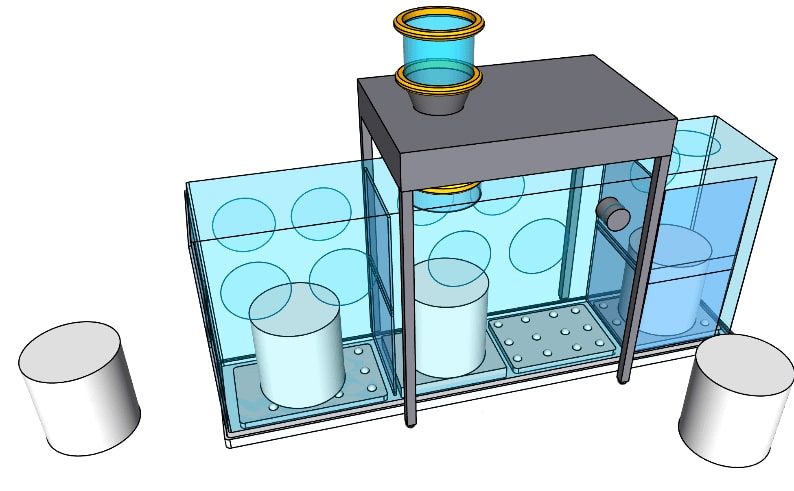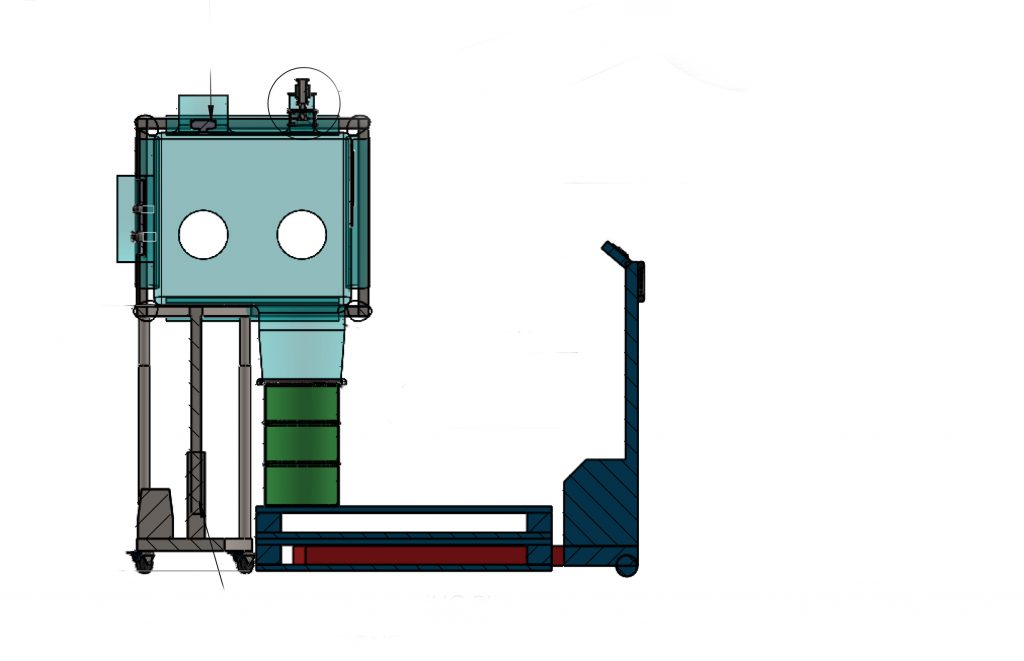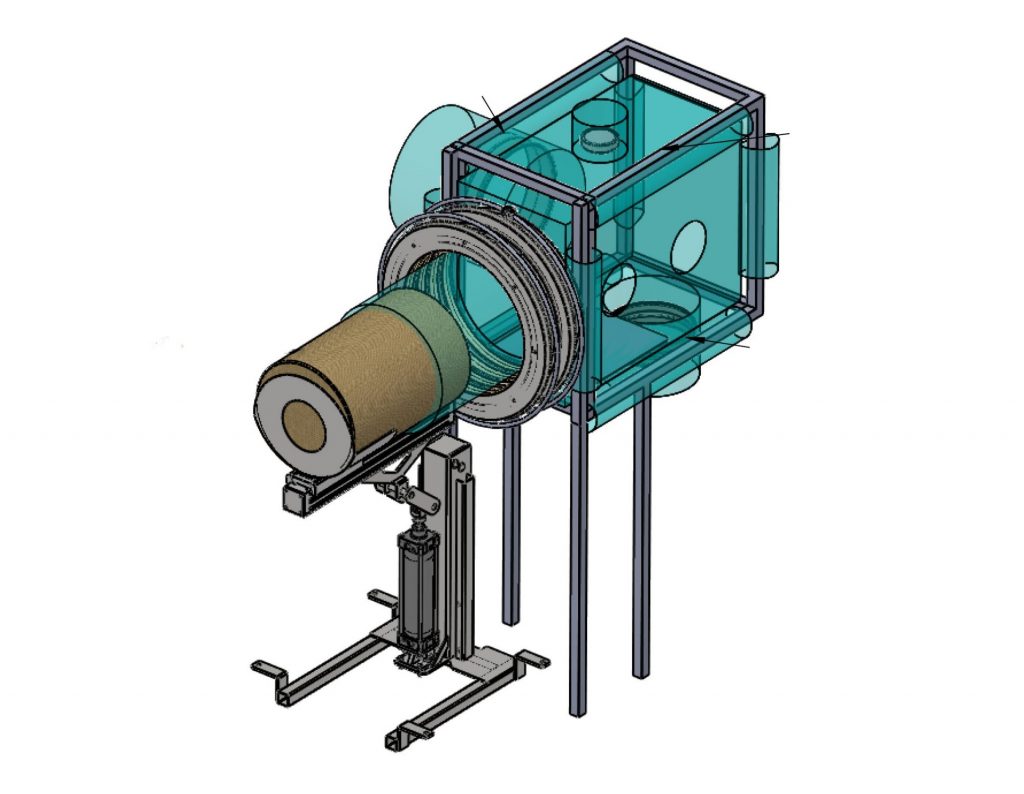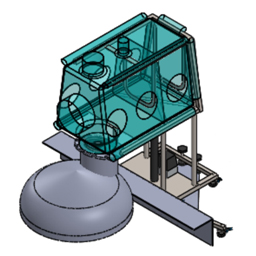
Flexible Isolators
In our manufacturing facility, classified as ISO 7/8, we produce, in accordance with GMP requirements and clients’ specific needs, continuous liners and flexible isolators, sometimes called glove bags, to confine all the exposed operations or temporarily modified the inner atmosphere (cleaner area, low humidity or inert needs).
Similar to plastic bubbles with gloves, filters, zippered doors, wires and hoses pass through and any required tools, they are usually ad hoc products. They are made of basic pharma low-density polyethylene or Polyurethene films directly supplied by CSV Containment, but we can also transform material provided by third parties indicated by the end customers.
The always-increasing quality of our work and the effort to improve constantly have led several costumers to authorize us as Qualified flexible technology supplier.
Flexible Pharma Solutions.
HPAPIFlexible Pharma Solutions for advanced containment and easy decontamination in pharmaceutical processes. Within the landscape of Flexible Pharma Solutions, the open-bottom flexible dispensing isolator represents one of the most interesting solutions in terms of decontamination efficiency and handling of highly potent APIs. Designed to ensure containment and ease of wiping, this system combines flexibility and […]
Custom Containment Solutions.
A personal custom containment expert does exist: with CSV, you can benefit from a truly customized containment service. Not just a single expert, but an entire team of technicians dedicated to your custom containment is the real promise of our offering for the pharmaceutical world and beyond.Do you need to develop a containment project where […]
Drum Sampling Automatic BIBO.
Why use a Bag In Bag Out interface for drum sampling? Drum sampling is a critical quality control operation in pharmaceutical manufacturing. It involves collecting representative samples of raw materials from drums while ensuring full protection for the operator, the product, and the surrounding environment. For this reason, drum sampling operations require a careful balance […]
Bag In Bag Out Automatic Interface.
Drum pneumatic powder transfer containment via the new Automatic Bag In Bag Out interface. CSV Containment has filed a patent application for an automatic interface designed to make coupling and powder transfer from drums via Bag In Bag Out safer, cleaner, and more efficient. Technique and technology. The now widespread and well-established Bag In Bag […]
How to contain a Roller Compactor.
The profile makes the difference, even when containing a pharmaceutical Roller Compactor. In a recent Roller Compactor upgrade project, we used a flexible isolator with already tested custom solutions, an approach that has now become a standard practice for us. The choice of a dedicated glove bag proved to be the ideal solution: easy to […]
HPAPI R&D Labs: Containing Potent Compounds.
HPAPI R&D environments are areas that require equal or greater attention than that given to production. When we talk about HPAPI R&D labs, we are referring to dynamic, evolving, and flexible environments, just like the solution that CSV Containment has designed for these types of activities. With a lower budget than rigid isolators, flexible technology […]
HPAPI Pilot Plant Upgrade
Is it possible to perform a pilot plant upgrade quickly and fully tailored to your needs? Our team of experts in flexible containment recently completed a pilot plant upgrade in Spain, working closely with the client throughout the entire process. Flexible containment technologies make it possible to achieve results that would otherwise require extensive facility […]
Flexible High Containment.
Is flexible high containment truly possible in terms of both material and operational versatility? Far from being an oxymoron, flexible high containment is a tangible solution that enables increasingly stringent safety standards in pharmaceutical manufacturing. The world of glove bags makes it possible to reduce both time and cost, offering the agility needed to stay […]
Instant Benchtop Isolator.
Csv Containment is launching the new, practical Instant Benchtop Isolator Single Use. The Instant Benchtop Isolator is the perfect solution in many situations, whether it’s quality control, laboratory testing or IPC. In the ever-changing pharmaceutical industry, you need quick and safe solutions. Drawing on its extensive experience in the field of flexible isolators, CSV Containment […]
DISPENSING GLOVE BAG for Every Need.
The Dispensing Glove Bag is the most requested flexible model, and CSV Containment responds to every request with tailor-made solutions. The Dispensing Glove Bag is the fastest and most practical solution for integrating or expanding a rapidly growing project, replacing an existing installation with a more cost-effective alternative, or investing in an R&D laboratory with […]
A Containment-proof Pharmaceutical Sieve.
A pharmaceutical sieve, properly contained, ensures that products are free of contamination and that operators can work safely. The pharmaceutical sieve plays an essential role in the production process of drugs. It can indeed protect products by avoiding contamination from both large particles (such as lumps, fibers, or threads) and anything else that might accidentally enter […]
OSD Manufacturing Facilities: a Flexible Upgrade.
OSD manufacturing facilities must ensure the highest levels of product quality and operator safety. The primary objective of international regulatory requirements for OSD manufacturing facilities is to protect pharmaceutical products and guarantee the highest product quality. Equally important, processes carried out within the facility must be streamlined and increasingly focused on safeguarding operator safety. CSV […]
Lenticular Filter Isolator.
We have enclosed a Lenticular Filter with flexible technology, creating a customised space tailored to meet the client’s specific needs. The Lenticular Filter is a piece of equipment that requires considerable maneuvering space, making containment operations a complex challenge. At CSV Containment, we enjoy taking on such challenges and, together with our client, explored the […]
Flexible Sampling and Dispensing Isolator.
Sampling and dispensing operations are more efficient with CSV Containment’s telescopic flexible isolators. Not only does the ergonomic design improve workflow, but safety is also enhanced through the negative pressure with a Venturi system. High containment performance ensures safe operations within the OEB4 band. Custom-made Solutions. All flexible isolators are manufactured in ISO8-classified production areas […]
HPAPI Flexible Containment for Pilot Plant.
HPAPI flexible containment solution for a fully contained pilot plant. HPAPI Flexible Containment was possible in a synthesis laboratory thanks to a modular structure with chambers integrated with each other. Our experience and ability to tailor work to handle almost any situation! Our CSV Containment teams always work in synergy with clients to find the […]
Flexible Room: a safe, swiftly constructed environment.
Thanks to our Flexible Rooms, it is possible to construct an environment capable of ensuring containment and safety in the typical case of an emergency production of a product at risk of contamination. What is a Flexible Room? It is a space equipped with an entry pass box, controlled ventilation, a tubular steel structure, and […]
Motorised Telescopic Glove Bag.
Our motorised telescopic glove bag is the answer to the difficulties of handling Bag in Bag Out drums under a flexible dispensing isolator workbench. Listening to manufacturing operators, at CSV Containment, we thought of a telescopic glove bag as a response to a concrete need. We have been challenged to avoid uncomfortable and very low […]
Centrifuge GMP temporary classified area.
Working Safely with a Centrifuge: A Tailored Challenge for CSV Containment. Creating a custom containment area around a centrifuge is made possible through the use of dedicated glove bags. The extensive experience gained in designing flexible isolators allows the technicians at CSV Containment to design ergonomic and safe structures for operators. Our containment experts also […]
Glove Bags for Reactor Loading.
More and more companies in the Pharma world are choosing glove bags for reactor loading, but what are the main advantages that this solution offers? Glove bags for reactor loading can guarantee a rapid response to the development needs of new processes in terms of costs and time. “We have tight schedules and within six […]
Safe Decontamination of Tools and Parts.
During the handling of powders, accessories may be used that need to be washed. But how can you obtain a safe decontamination of tools and parts in the same environment, while taking up minimal space? CSV Containment has leveraged its expertise in creating custom flexible isolators to allow operators handling powders to get a safe […]
Flexible weighing and dispensing isolators.
The range of flexible weighing and dispensing isolators by CSV Containment ensures containment levels up to OEB 5 and can be equipped with tools for controlling the internal atmosphere to operate under negative pressure, controlled humidity, and/or inert environment. Each of the flexible weighing and dispensing isolators made by CSV Containment is customizable according to the […]
ECP: from toxicology to a Containment Strategy
A proper containment strategy can be implemented, starting with a toxicology profile assessment performed by professionals in a pharmaceutical facility. OEL values (Occupational exposure limits derived from toxicological data) are used by pharmaceutical industries as a starting point to determine the containment strategy to apply, in the case of both a new facility and an […]
Pneumatic powder transfer from drums: a GMP dust-free solution!
Watch the video! A flexible system to confine pneumatic powder transfer operations, not only because of the material of the isolator but also because of the flexibility in serving drums of different diameters. Frequently, pneumatic systems operating in vacuum are used for pneumatic powder transfer operations, mainly to avoid handling drums and containers at height […]
SMEPAC Monitoring.
Containment and capability: two words that tell the story of CSV Containment’s world, but they also summarize the meaning of SMEPAC monitoring, aimed at verifying the safety of operators and equipment. Indeed, SMEPAC monitoring (Standardized Measurement of Equipment Particulate Airborne Concentration) is aimed at measuring the particulate containment performance of isolation systems used in the pharmaceutical […]
Tray and Freeze Dryer Containment.
Tray and freeze dryer Containment via flexible isolators is the CSV solution when there are no alternatives to ensure operators and production safety. The tray and freeze dryer Containment is an example of how our flexible isolators can adapt to innovative and unusual situations and solve otherwise unmanageable problems. Bringing existing equipment to a containment […]
DIT® Flexible Isolators.
DIT® Flexible Isolators’ technology represents one of the most versatile solutions on the market for safe reactor charging. DIT® flexible isolators are the result of combining two experiences in which CSV Containment is achieving important results: materials processed in the “Flexilab” and patented iris technology. The materials, which are certified and produced in compliance with […]
Realizing custom flexible isolators in a short time: this is our mission!
A success story: the realization of Containment Flexible Isolators for an R&D department dedicated to the Pharmaceutical Technology of Oral Solid Products. CSV Containment isolators are proving to be more and more flexible, not only in terms of material but also in terms of how, where, and when they are used. The “how” is experienced […]
Flexible isolators for OSD facility: all CSV Containment services and solutions.
As in the case of OSD facility, the flexibility of flexibles it is not simply a play on words, but a feature that CSV Containment knows how to make the most of. In order to assure a containment performance in an OSD facility, within OEB4 and OEB5 classes, CSV Containment has faced a particular request […]
Contained Unloading System for a Rotating dryer trough a mill.
The main trait of Csv Containment is to find optimal containment solutions for every situation, even for pre-existing ones, as in the case of this Contained unloading system placed under a mill. Contained unloading system means assuring safe operation during a specific processing phase. In addition to this basic goal, having an overall view of […]
QC Lab Flexible Isolators: a focus on laboratory safety and quality control
Our QC lab flexible isolators are designed with you in mind to improve your work safety and quality. But, how can you increase work safety through upgrades that still guarantee process quality standards? Laboratory quality control (QC) is a necessary activity to eliminate the risk of non-compliant outcomes. Indeed, lab QC ensures that all the […]
Universal Lifters for Drums.
CSV Containment realizes telescopic universal lifters for drums to meet the different conditions of work environments. Universal lifters allow bringing the drum as close as possible to the charging point thanks to both the telescopic structure and the compass-opening legs. The lifters are movable and have braked, pivoting, and antistatic wheels. Drums of different sizes […]
Disposable and single use Split Valve for Reactor Charging and Sampling.
A new solution for Reactor Charging and Sampling through Disposable and Single Use Split Valve on Glove Bags, Charge Bags and Sampling Sleeves. Disposable Split Valve on Glove Bags: our charging solution is made of a clear and solvent resistant flexible isolator (antistatic PU or LDPE at choice), equipped with: 2 gloves a DN350 bag […]
Universal flexible dispensing skid.
New materials to tailor your flexible dispensing isolator and increase the safety. CSV Containment displays a universal dispensing isolator to improve the efficiency of your production processes in areas where highly active substances are handled. The disposable technologies can protect both the operator and the product during the filling, unloading, weighing and sampling of substances. The […]
Glass Reactor Manway ISOFLEX.
A new universal solution for Glass Reactor Charging. Whatch the video tutorial The glass reactor charging point is a manway, usually a PTFE cap, modified with a 4” o a 6” end clamp that host an active EZIFLOW cassette. Here some pictures showing : the original Glass Reactor PTFE manway the end clamp modified PTFE manway […]
A performance isolator test to upgrade standard configuration machines.
CSV Containment offer a service of isolator test during the construction phase. These validation isolator test on flexible solutions are carried out by analyzing any leaks with the introduction of technical gas.The flexible enclosure is installed on the base frame of the machine, thus creating a volume suitable to be filled with helium gas, with very high […]
Drum Sampling Flexible Containment
A mobile drum sampling skid to work in safety. CSV Containment presents a drum sampling isolator made by super transparent TPU film. As you can see in the video, working with this film in a trasfer powder operation is easier. The station is now available with a swinging steel made grooved canister and equipped with new […]
Drum to Drum Weighing adjustment station with super transparent film.
A new super transparent film for any flexible skid. CSV Containment continues its research to improve the new isolators with the introduction of a new super transparent film. As you can see in the video, working with this film in a trasfer powder operation is easier. The drum to drum weighing station is now available […]
Flexible isolators made in Italy, thinked for greater operator comfort.
A different way of viewing the Containment with our new flexible isolators. CSV Containment reinforces its position in the production of flexible isolators and containment systems investing in research and development. A marked versatility has allowed CSV Containment to create different solutions of Insulators and Glove Bags with particular shapes and colors, aimed at confining the most […]
Filter cake recovery isolator for openable dryer base
Contained filter cake recovery solution for openable dryer base made by Csv Containment. Filter cake recovery and cleaning operations are fundamental stages in the production processes of the pharmaceutical and biotechnological sectors. Oftentimes, though, when dealing with toxic substances, filter-dryer discharge operation can become a complex and dangerous task. CSV Containment offers the ideal solution […]
Filter dryer bottom valve cleaning and cake recovery
Cleaning solution for filter dryer bottom valve made by Csv Containment. Residual product recovery and discharge valve cleaning operations are fundamental stages in the production processes of the pharmaceutical and biotechnological sectors. Oftentimes, though, when dealing with toxic substances, even a cake recovery and filter-dryer discharge operation can become a complex and dangerous task. CSV […]
Before Smepac
A performance test in the production phase of isolators, repeatable on site post installation. Safety is an essential issue during the production process and we all know the value of SMEPAC test. CSV Contaimnent wants to go further and offer a service to test the isolator during the realization phase. These validation tests on […]
Liquid Drum Transfer Containment
Pneumatic Transfer Shuttle Pneumatic Drum Lifting shuttle to lift up and down the chamber together with the grooved canister, surrounding the drum and allowing an ergonomic lance safe extraction. Features of the CSV Pneumatic Transfer skid: Single Chamber Flexible Drum Transfer Isolator 2 Gloves Steel support structure (AISI 304) Steel (Bag In Bag Out) Bottom […]
Rotating dryer charging skid & flexible isolator
Dryer drum elevator charging The rotating dryer charging trolley, allows to get a sturdy automatic coupling between the drum and the dyrer, by means of a pneumatic lifter, thus enabling a safety charge and discharge of the product, through a decidated temporary flexible chamber. A clamping connection ensure the skid to the structure of the […]
Iris Technology – Weight Adjustment Station Drum to Drum
New Flexible Weight Adjustment Station Weight Adjustement Station is a Flexible Isolator equipped by Iris Technology The station features: Antistatic Pharmaceutical Solvent compatible LDPE ATEX version available Two sides operations (2 gloves each side) N2 injection available As optional a negative pressure Venturi device can fit the support structure Zippered wall to enter clean goods Two pneumatic Iris made […]
Drum Liquid Dispensing Flexible Isolator by CSV Containment
CSV Containment is proud to present its Drum Liquid Dispensing Flexible Isolator Drum Liquid Dispensing Flexible Skid is an Hybrid Equipment made of one telescopic and movable support skid and one Drum Flexible Isolator The skid features: Movable (pivotal wheels with breaks) Telescopic (by manual Hydraulic lifting piston) Antistatic Pharmaceutical Solvent compatible LDPE ATEX version available Excellent Ergonomic feeling thanks to its Trapezoidal […]
CSV Containment – DRUM TRANSFER FLEXIBLE SKID
CSV Containment – Drum Transfer Flexible Skid Proud to introduce a new loading station for dryers or other equipments loaded from drums. The loading station is mobile and telescopic with a manual hydraulic mechanism that works on braked and pivoting wheels. The isolation chamber for the unloading drum operations is flexible and equipped with two gloves, […]
CSV Containment – Freeze Dryer Flexible Containment
CSV Containment – Freeze Dryer Flexible Containment CSV flexible solution for freeze drying containment Features: Triple chamber (pre chamber, main & offloading chamber) IN/OUT material flow separation Negative pressure cascade control Flexible zippered door for isolator/freeze dryer safe detaching No steel surfaces product contact Fully ergonomic sliding/swinging freeze dryer offloading chamber working bench If […]
Flexible Isolator washing in place?
Polyethylene (LDPE) with a good compatibility rating with most chemicals, resistance to solvents maintaining physical properties; Disposable item using the same product (campaigns); Solutions adapted to each production process. Watch the video! CSV Containment is present in the pharmaceutical market and is focused on providing engineering, construction and validation solutions for the LifeScience world. For more information:containment@csv-ls.com
CSV Containment is proud to present its IRIS BIG BAG Interface!
CSV Containment is proud to present its ! This new device includes a few innovations and has been developed to guarantee ever-increasing quality/containment for CSV customers. A FLEXIBLE ISOLATOR allows to contain big bag coupling and detaching operations; A top IRIS squeezes the big bag offloading neck and allows powder flow dosing; Easily Upgradable to High Containment Configuration […]
CSV Containment presents Flexible Battery for Filters Safe Change
The Isoflex has the following features: Zippered material pass box sleeve to clean filters transfer in; Waste Bag out for dirty Filters, via continuous Liner; N2 Injection, Spray gun and draining bottom point; Easy to install and to be collapsed for incineration; Solvents compatible; ATEX rated. CSV Containment is present in the pharmaceutical market and is focused on […]
CSV Containment presents
Manway Reactor Charging Flexible Isolator
The Manway Charging Isoflex has the following features: Zippered conical neck to be secured around the closed manway Easy installation around the (Still closed) manway Manway opening in closed/contained atmosphere Zippered/weloc Material air lock to transfer in any Material/tools Waste Bag out Liner N2 Injection, Spray gun and draining bottom point Solvents compatible ATEX certified […]
CSV Containment introduces the Dispensing Flexible Isolator
The Dispensing Flexible Isolator, developed by CSV Containment, is a new containment technology, that has the following characteristics: Compact and movable Plug and play utilities Collapsible frame for storage Designed for any discharging interfaces/containers Option with ventilation available No sw automation required for pressure control Suitable for high containment CSV Containment is present in the pharmaceutical market […]